 Hideaki Nagase, Ph.D.
Hideaki Nagase, Ph.D.
Kennedy Institute of Rheumatology-Centre for Degenerative Diseases, University of Oxford, UK
Osteoarthritis: diagnosis, treatment and challenges
Hideaki Nagase1, Ngee Han Lim1, George Bou-Gharios1, Ernst Meinjohanns2 and Morten Meldal3
- Kennedy Institute of Rheumatology, Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences, University of Oxford, London, W6 8LH UK
- Carlsberg Laboratory, Copenhagen, Denmark,
- Nano-Science Center, Department of Chemistry, University of Copenhagen, Denmark
Osteoarthritis (OA) is the most prevalent age-related degenerative joint disease. With the expanding ageing population, it imposes a major socio-economic burden on society. A key feature of OA is a gradual loss of articular cartilage and deformation of bone, resulting in the impairment of joint function. Currently, there is no effective disease-modifying treatment except joint replacement surgery. There are many possible causes of cartilage loss (e.g. mechanical load, injury, reactive oxygen species, aging, etc.) and etiological factors (obesity, genetics), but the degradation of cartilage is primarily caused by elevated levels of active metalloproteinases. It is therefore attractive to consider proteinase inhibitors as potential therapeutics. However, there are several hurdles to overcome, namely early diagnosis and continuous monitoring of the efficacy of inhibitor therapeutics. We are therefore aiming at developing non-invasive probes to detect cartilage degrading metalloproteinase activities.
We have designed in vivo imaging probes to detect MMP-13 (collagenase 3) activity that participates in OA by degrade cartilage collagen II and MMP-12 (macrophage elastase) activity involved in inflammatory arthritis. These activity-based probes consist of a peptide that is selectively cleaved by the target proteinase, a near-infrared fluorophore and a quencher. The probe’s signal multiplies upon proteolysis. They were first used to follow the respective enzyme activity in vivo in the mouse model of collagen-induced arthritis and we found MMP-12 activity probe (MMP12AP) activation peaked at 5 days after onset of the disease, whereas MMP13AP activation was observed at 10-15 days. The in vivo activation of these probes was inhibited by specific low molecule inhibitors. We proceeded to test both probes in the mouse model of OA induced by the surgical destabilization of medial meniscus of the knee joints. In this model, degradation of knee cartilage is first detected histologically 6 weeks after surgery with significant erosion detectable at 8 weeks. Little activation of MMP12AP was detected, which was expected, as macrophage migration is not obvious in OA. MMP13AP, on the other hand, was significantly activated in the operated knee at 6 weeks compared with the non-operated contralateral knee, but there were no significant differences between the operated and sham-operated knees. At 8 weeks, however, the signals in the operated knees were significantly higher than both the contralateral and sham-operated controls. Activation of aggrecanases and MMP-13 are observed before structural changes of cartilage. We are therefore currently improving the MMP-13 probe for earlier detection by attaching it to polymers that are retained in cartilage.
 Gillian Murphy, Ph.D.
Gillian Murphy, Ph.D.
Professor, Department of Oncology, University of Cambridge, UK
A novel strategy for targeting metalloproteinases in cancer
Epithelial tumours evolve in a multi-step manner, involving both inflammatory and mesenchymal cells. Although intrinsic factors drive malignant progression, the influence of the micro-environment of neoplastic cells is a major feature of tumorigenesis. Extracellular proteinases, notably the metalloproteinases, are key players in the regulation of this cellular environment, acting as major effectors of both cell-cell and cell-extracellular matrix (ECM) interactions. They are involved in modifying ECM integrity, growth factor availability and the function of cell surface signalling systems, with consequent effects on cellular differentiation, proliferation and apoptosis.This has made metalloproteinases important targets for therapeutic interventions in cancer and small molecule inhibitors focussed on chelation of the active site zinc and binding within the immediate active site pocket were developed. These were not successful in early clinical trials due to the relative lack of specificity and precise knowledge of the target proteinase(s) in specific cancers. We can now appreciate that it is essential that we understand the relative roles of the different enzymes (of which there are over 60) in terms of their pro and anti tumour activity and their precise sites of expression The next generations of metalloproteinase inhibitors need the added specificity that might be gained from an understanding of the structure of individual active sites and the role of extra catalytic domains in substrate binding and other aspects of their biology. We have prepared scFv antibodies to the extra catalytic domains of two membrane metalloproteinases, MMP-14 and ADAM17, that play key roles in the tumour microenvironment. Our rationale and experiences with these agents will be presented in more detail.
 Colin Barrow, Ph.D.
Colin Barrow, Ph.D.
Chair in Biotechnology, School of Life & Environmental Sciences, Deakin University, Australia
Nano-biotechnology: Omega-3 Oils and Nanofibres
The health benefits of long-chain omega-3 fatty acids are well established, especially for eicosapentaenoic acid (EPA) and docosapentaenoic acid (DHA) from fish and microbial sources. In fact, a billion dollar market exists for these compounds as nutritional supplements, functional foods and pharmaceuticals. This presentation will describe some aspects of our omega-3 biotechnology research that are at the intersection of Nano-biotechnology and oil chemistry. These include the use of lipases for the concentration of omega-3 fats, through immobilization of these lipases on nanoparticles, and the microencapsulation and stabilization of omega-3 oils for functional foods. I will also describe some of our work on the enzymatic production of resolvins using lipoxygenases, and the fermentation of omega-3 oils from marine micro-organisms. Finally, I will describe some of our work on the formation of amyloid fibrils and graphene for various applications in nano-biotechnology.
 Sanjeeva Srivastava, Ph.D.
Sanjeeva Srivastava, Ph.D.
Assistant Professor, Proteomics Lab, IIT-Bombay, India
Identification of Potential Early Diagnostic Biomarkers for Gliomas and Various Infectious Diseases using Proteomic Technologies
The spectacular advancements achieved in the field of proteomics research during the last decade have propelled the growth of proteomics for clinical research. Recently, comprehensive proteomic analyses of different biological samples such as serum or plasma, tissue, CSF, urine, saliva etc. have attracted considerable attention for the identification of protein biomarkers as early detection surrogates for diseases (Ray et al., 2011). Biomarkers are biomolecules that can be used for early disease detection, differentiation between closely related diseases with similar clinical manifestations as well as aid in scrutinizing disease progression. Our research group is performing in-depth analysis of alteration in human proteome in different types of brain tumors and various pathogenic infections to obtain mechanistic insight about the disease pathogenesis and host immune responses, and identification of surrogate protein markers for these fatal human diseases.
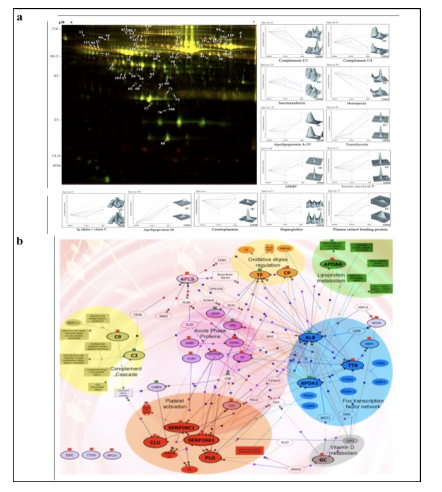
Applying 2D-DIGE in combination with MALDI-TOF/TOF MS we have analyzed the serum and tissue proteome profiles of glioblastoma multiforme; the most common and lethal adult malignant brain tumor (Gollapalli et al., 2012) (Figure 1). Results obtained were validated by employing different immunoassay-based approaches. In serum proteomic analysis we have identified some interesting proteins like haptoglobin, ceruloplasmin, vitamin-D binding protein etc. Moreover, proteomic analysis of different grades (grade-I to IV) of gliomas and normal brain tissue was performed and differential expressions of quite a few proteins such as SIRT2, GFAP, SOD, CDC42 have been identified, which have significant correlation with the tumor growth. While proteomic analysis of cerebrospinal fluid from low grade (grade I & II) vs. high grade (grade III & IV) gliomas revealed modulation of CSF levels of apolipoprotein E, dickkopf related protein 3, vitamin D binding protein and albumin in high grade gliomas. The prospective candidates identified in our studies provide a mechanistic insight of glioma pathogenesis and identification of potential biomarkers. We are also studying the role of JAK/STAT interactome and therapeutic potential of STAT3 inhibitors in gliomas using proteomics approach. Several candidates of the JAK/STAT interactome were identified with altered expression and a significant correlation was observed between STAT3 and PDK1 transcript expression level.
We have also investigated the changes in human serum proteome in different infectious diseases including falciparum and vivax malaria (Ray et al., 2012a; Ray et al., 2012b), dengue (Ray et al., 2012c) and leptospirosis (Srivastava et al., 2012). Although, quite a few serum proteins were found to be commonly altered in different infectious diseases and might be a consequence of inflammation mediated acute phase response signaling, uniquely modulated candidates were identified in each pathogenic infection indicating the some inimitable responses. Further, a panel of identified proteins consists of six candidates; serum amyloid A, hemopexin, apolipoprotein E, haptoglobin, retinol-binding protein and apolipoprotein A-I was used to build statistical sample class prediction models employing PLSDA and other classification methods to predict the clinical phenotypic classes and 91.37% overall prediction accuracy was achieved (Figure 2). ROC curve analysis was carried out to evaluate the individual performance of classifier proteins. The excellent discrimination among the different disease groups on the basis of differentially expressed proteins demonstrates the potential diagnostic implications of this analytical approach.
Keywords: Diagnostic biomarkers, Gliomas, Infectious Diseases, Proteomics, Serum proteome
Acknowledgments: This disease biomarker discovery research was supported by Department of Biotechnology, India grant (No. BT/PR14359/MED/30/916/2010), Board of Research in Nuclear Sciences (BRNS) DAE young scientist award (2009/20/37/4/BRNS) and a startup grant 09IRCC007 from the IIT Bombay. The active support from Advanced Center for Treatment Research and Education in Cancer (ACTREC), Tata Memorial Hospital (TMH), and Seth GS Medical College and KEM Hospital Mumbai, India in clinical sample collection process is gratefully acknowledged.
References :
- Ray S, Reddy PJ, Jain R, Gollapalli K. Moiyadi A, Srivastava S. Proteomic technologies for the identification of disease biomarkers in serum: advances and challenges ahead. Proteomics 11: 2139-61, 2011.
- Gollapalli K, Ray S, Srivastava R, Renu D, Singh P, Dhali S, Dikshit JB, Srikanth R, Moiyadi A, Srivastava S. Investigation of serum proteome alterations in human glioblastoma multiforme. Proteomics 12(14): 2378-90, 2012.
- Ray S, Renu D, Srivastava R, Gollapalli K, Taur S, Jhaveri T, Dhali S, Chennareddy S, Potla A, Dikshit JB, Srikanth R, Gogtay N, Thatte U, Patankar S, Srivastava S. Proteomic investigation of falciparum and vivax malaria for identification of surrogate protein markers. PLoS One 7(8): e41751, 2012a.
- Ray S, Kamath KS, Srivastava R, Raghu D, Gollapalli K, Jain R, Gupta SV, Ray S, Taur S, Dhali S, Gogtay N, Thatte U, Srikanth R, Patankar S, Srivastava S. Serum proteome analysis of vivax malaria: An insight into the disease pathogenesis and host immune response. J Proteomics 75(10): 3063-80, 2012b.
- Srivastava R, Ray S, Vaibhav V, Gollapalli K, Jhaveri T, Taur S, Dhali S, Gogtay N, Thatte U, Srikanth R, Srivastava S. Serum profiling of leptospirosis patients to investigate proteomic alterations. J Proteomics 76: 56-68, 2012.
- Ray S, Srivastava R, Tripathi K, Vaibhav V, Srivastava S. Serum proteome changes in dengue virus-infected patients from a dengue-endemic area of India: towards new molecular targets? OMICS 16(10): 527-36, 2012c.
* Correspondence: Dr. Sanjeeva Srivastava, Department of Biosciences and Bioengineering, IIT Bombay, Mumbai 400 076, India: E-mail: sanjeeva@iitb.ac.in; Phone: +91-22-2576-7779, Fax: +91-22-2572-3480
![Figure 2 (a) Western blot analysis of haptoglobin (HP), serum amyloid A (SAA), and clusterin (CLU) from serum samples of healthy control (HC) [n = 12], falciparum malaria (FM) [n = 12], vivax malaria (VM) [n = 12], Leptospirosis (Lep) [n = 6], dengue fever [DF] [n = 6] and non infectious disease control (NIDC:GBM) [n = 12]. Representative blots of the target proteins are depicted along with their respective relative abundance volumes (volume X 104). All the data are represented as mean ± SE. (b) Discrimination of malaria from dengue, leptospirosis and GBM using PLS-DA analysis. PLS-DA scores Plot for FM (blue spheres, n = 8), VM (green spheres, n = 8), DF (red spheres, n = 6), Lep (grey spheres, n = 6) and GBM (brown spheres, n = 8) samples based on 6 differentially expressed proteins (serum amyloid A, hemopexin, apolipoprotein E, haptoglobin, retinol-binding protein and apolipoprotein A-I) identified using DIGE. The axes of the plot indicate PLSDA latent variables t0-t2.](http://www.amritabioquest.org/conference/2013/wp-content/uploads/sites/2/2015/02/sanjeeva-2.jpg)
 Manzoor K, Ph.D.
Manzoor K, Ph.D.
Professor, Centre for Nanoscience & Molecular Medicine, Amrita University
Targeting aberrant cancer kinome using rationally designed nano-polypharmaceutics
Manzoor Koyakutty, Archana Ratnakumary, Parwathy Chandran, Anusha Ashokan, and Shanti Nair
`War on Cancer’ was declared nearly 40 years ago. Since then, we made significant progress on fundamental understanding of cancer and developed novel therapeutics to deal with the most complex disease human race ever faced with. However, even today, cancer remains to be the unconquered `emperor of all maladies’. It is well accepted that meaningful progress in the fight against cancer is possible only with in-depth understanding on the molecular mechanisms that drives its swift and dynamic progression. During the last decade, emerging new technologies such as nanomedicine could offer refreshing life to the `war on cancer’ by way of providing novel methods for molecular diagnosis and therapy.
In the present talk, we discuss our approaches to target critically aberrant cancer kinases using rationally designed polymer-protein and protein-protein core-shell nanomedicines. We have used both genomic and proteomic approaches to identify many intimately cross-linked and complex aberrant protein kinases behind the drug resistance and uncontrolled proliferation of refractory leukemic cells derived from patients. Small molecule inhibitors targeted against oncogenic pathways in these cells were found ineffective due to the involvement of alternative survival pathways. This demands simultaneous inhibition more than one oncogenic kinases using poly-pharmaceutics approach. For this, we have rationally designed core-shell nanomedicines that can deliver several small molecules together for targeting multiple cancer signalling. We have also used combination of small molecules and siRNA for combined gene silencing together with protein kinase inhibition in refractory cancer cells. Optimized nanomedicines were successfully tested in patient samples and found enhanced cytotoxicity and molecular specificity in drug resistant cases.
Nano-polypharmaceutics represents a new generation of nanomedicines that can tackle multiple cancer mechanisms simultaneously. Considering the complexity of the disease, such therapeutic approaches are not simply an advantage, but indispensable.
Acknowledgements:
We thank Dept. of Biotechnology and Dept. Of Science and Technology,Govt. of India for the financial support through `Thematic unit of Excellence in Medical NanoBiotechnology’ and `Nanomedicine- RNAi programs’.

Syed Salman Lateef and Vinayak A K
Development of Supercritical Fluid Chromatography methods for the replacement of existing USP Normal phase liquid chromatography methods
Normal phase liquid chromatography methods often have long run times and involve environmentally toxic/costly solvents. Supercritical chromatography methods on the other hand are faster, inexpensive, and eco-friendly. The low viscous supercritical carbon dioxide operates at high flow rates compared to LC without losing separation efficiency. In this work, SFC methods are developed to replace three United States Pharmacopeial (USP) normal phase achiral methods – prednisolone, tolazamide and cholecalciferol. System suitability parameters of the normal phase method are compared against the SFC method. Precision, linearity and robustness of the new SFC methods are demonstrated. SFC methods were found to be cost effective in terms of analysis time and solvent savings. The SFC method does not require purchase and disposal of expensive environmentally hazardous chemicals. Hence, the newly developed SFC method provides a faster and safer solution.




