 Rohit Manchanda, Ph.D.
Rohit Manchanda, Ph.D.
Professor, Biomedical Engineering Group, IIT-Bombay, India
Modelling the syncytial organization and neural control of smooth muscle: insights into autonomic physiology and pharmacology
We have been studying computationally the syncytial organization and neural control of smooth muscle in order to help explain certain puzzling findings thrown up by experimental work. This relates in particular to electrical signals generated in smooth muscles, such as synaptic potentials and spikes, and how these are explicable only if three-dimensional syncytial biophysics are taken fully into account. In this talk, I shall provide an illustration of outcomes and insights gleaned from such an approach. I shall first describe our work on the mammalian vas deferens, in which an analysis of the effects of syncytial coupling led us to conclude that the experimental effects of a presumptive gap junction uncoupler, heptanol, on synaptic potentials were incompatible with gap junctional block and could best be explained by a heptanol-induced inhibition of neurotransmitter release, thus compelling a reinterpretation of the mechanism of action of this agent. I shall outline the various lines of evidence, based on indices of syncytial function, that we adduced in order to reach this conclusion. We have now moved on to our current focus on urinary bladder biophysics, where the questions we aim to address are to do with mechanisms of spike generation. Smooth muscle cells in the bladder exhibit spontaneous spiking and spikes occur in a variety of distinct shapes, making their generation problematic to explain. We believe that the variety in shapes may owe less to intrinsic differences in spike mechanism (i.e., in the complement of ion channels participating in spike production) and more to features imposed by syncytial biophysics. We focus especially on the modulation of spike shape in a 3-D coupled network by such factors as innervation pattern, propagation in a syncytium, electrically finite bundles within and between which the spikes spread, and some degree of pacemaker activity by a sub-population of the cells. I shall report two streams of work that we have done, and the tentative conclusions these have enabled us to reach: (a) using the NEURON environment, to construct the smooth muscle syncytium and endow it with synaptic drive, and (b) using signal-processing approaches, towards sorting and classifying the experimentally recorded spikes.
 Sanjeeva Srivastava, Ph.D.
Sanjeeva Srivastava, Ph.D.
Assistant Professor, Proteomics Lab, IIT-Bombay, India
Identification of Potential Early Diagnostic Biomarkers for Gliomas and Various Infectious Diseases using Proteomic Technologies
The spectacular advancements achieved in the field of proteomics research during the last decade have propelled the growth of proteomics for clinical research. Recently, comprehensive proteomic analyses of different biological samples such as serum or plasma, tissue, CSF, urine, saliva etc. have attracted considerable attention for the identification of protein biomarkers as early detection surrogates for diseases (Ray et al., 2011). Biomarkers are biomolecules that can be used for early disease detection, differentiation between closely related diseases with similar clinical manifestations as well as aid in scrutinizing disease progression. Our research group is performing in-depth analysis of alteration in human proteome in different types of brain tumors and various pathogenic infections to obtain mechanistic insight about the disease pathogenesis and host immune responses, and identification of surrogate protein markers for these fatal human diseases.
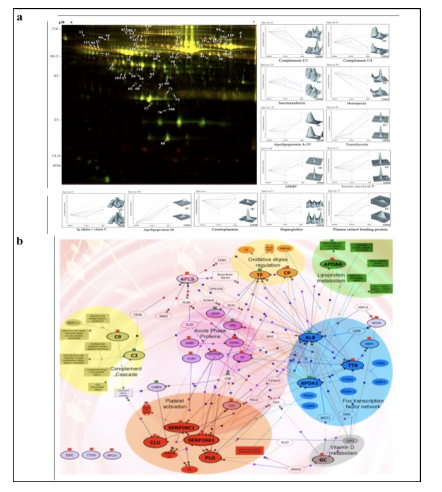
Applying 2D-DIGE in combination with MALDI-TOF/TOF MS we have analyzed the serum and tissue proteome profiles of glioblastoma multiforme; the most common and lethal adult malignant brain tumor (Gollapalli et al., 2012) (Figure 1). Results obtained were validated by employing different immunoassay-based approaches. In serum proteomic analysis we have identified some interesting proteins like haptoglobin, ceruloplasmin, vitamin-D binding protein etc. Moreover, proteomic analysis of different grades (grade-I to IV) of gliomas and normal brain tissue was performed and differential expressions of quite a few proteins such as SIRT2, GFAP, SOD, CDC42 have been identified, which have significant correlation with the tumor growth. While proteomic analysis of cerebrospinal fluid from low grade (grade I & II) vs. high grade (grade III & IV) gliomas revealed modulation of CSF levels of apolipoprotein E, dickkopf related protein 3, vitamin D binding protein and albumin in high grade gliomas. The prospective candidates identified in our studies provide a mechanistic insight of glioma pathogenesis and identification of potential biomarkers. We are also studying the role of JAK/STAT interactome and therapeutic potential of STAT3 inhibitors in gliomas using proteomics approach. Several candidates of the JAK/STAT interactome were identified with altered expression and a significant correlation was observed between STAT3 and PDK1 transcript expression level.
We have also investigated the changes in human serum proteome in different infectious diseases including falciparum and vivax malaria (Ray et al., 2012a; Ray et al., 2012b), dengue (Ray et al., 2012c) and leptospirosis (Srivastava et al., 2012). Although, quite a few serum proteins were found to be commonly altered in different infectious diseases and might be a consequence of inflammation mediated acute phase response signaling, uniquely modulated candidates were identified in each pathogenic infection indicating the some inimitable responses. Further, a panel of identified proteins consists of six candidates; serum amyloid A, hemopexin, apolipoprotein E, haptoglobin, retinol-binding protein and apolipoprotein A-I was used to build statistical sample class prediction models employing PLSDA and other classification methods to predict the clinical phenotypic classes and 91.37% overall prediction accuracy was achieved (Figure 2). ROC curve analysis was carried out to evaluate the individual performance of classifier proteins. The excellent discrimination among the different disease groups on the basis of differentially expressed proteins demonstrates the potential diagnostic implications of this analytical approach.
Keywords: Diagnostic biomarkers, Gliomas, Infectious Diseases, Proteomics, Serum proteome
Acknowledgments: This disease biomarker discovery research was supported by Department of Biotechnology, India grant (No. BT/PR14359/MED/30/916/2010), Board of Research in Nuclear Sciences (BRNS) DAE young scientist award (2009/20/37/4/BRNS) and a startup grant 09IRCC007 from the IIT Bombay. The active support from Advanced Center for Treatment Research and Education in Cancer (ACTREC), Tata Memorial Hospital (TMH), and Seth GS Medical College and KEM Hospital Mumbai, India in clinical sample collection process is gratefully acknowledged.
References :
- Ray S, Reddy PJ, Jain R, Gollapalli K. Moiyadi A, Srivastava S. Proteomic technologies for the identification of disease biomarkers in serum: advances and challenges ahead. Proteomics 11: 2139-61, 2011.
- Gollapalli K, Ray S, Srivastava R, Renu D, Singh P, Dhali S, Dikshit JB, Srikanth R, Moiyadi A, Srivastava S. Investigation of serum proteome alterations in human glioblastoma multiforme. Proteomics 12(14): 2378-90, 2012.
- Ray S, Renu D, Srivastava R, Gollapalli K, Taur S, Jhaveri T, Dhali S, Chennareddy S, Potla A, Dikshit JB, Srikanth R, Gogtay N, Thatte U, Patankar S, Srivastava S. Proteomic investigation of falciparum and vivax malaria for identification of surrogate protein markers. PLoS One 7(8): e41751, 2012a.
- Ray S, Kamath KS, Srivastava R, Raghu D, Gollapalli K, Jain R, Gupta SV, Ray S, Taur S, Dhali S, Gogtay N, Thatte U, Srikanth R, Patankar S, Srivastava S. Serum proteome analysis of vivax malaria: An insight into the disease pathogenesis and host immune response. J Proteomics 75(10): 3063-80, 2012b.
- Srivastava R, Ray S, Vaibhav V, Gollapalli K, Jhaveri T, Taur S, Dhali S, Gogtay N, Thatte U, Srikanth R, Srivastava S. Serum profiling of leptospirosis patients to investigate proteomic alterations. J Proteomics 76: 56-68, 2012.
- Ray S, Srivastava R, Tripathi K, Vaibhav V, Srivastava S. Serum proteome changes in dengue virus-infected patients from a dengue-endemic area of India: towards new molecular targets? OMICS 16(10): 527-36, 2012c.
* Correspondence: Dr. Sanjeeva Srivastava, Department of Biosciences and Bioengineering, IIT Bombay, Mumbai 400 076, India: E-mail: sanjeeva@iitb.ac.in; Phone: +91-22-2576-7779, Fax: +91-22-2572-3480
![Figure 2 (a) Western blot analysis of haptoglobin (HP), serum amyloid A (SAA), and clusterin (CLU) from serum samples of healthy control (HC) [n = 12], falciparum malaria (FM) [n = 12], vivax malaria (VM) [n = 12], Leptospirosis (Lep) [n = 6], dengue fever [DF] [n = 6] and non infectious disease control (NIDC:GBM) [n = 12]. Representative blots of the target proteins are depicted along with their respective relative abundance volumes (volume X 104). All the data are represented as mean ± SE. (b) Discrimination of malaria from dengue, leptospirosis and GBM using PLS-DA analysis. PLS-DA scores Plot for FM (blue spheres, n = 8), VM (green spheres, n = 8), DF (red spheres, n = 6), Lep (grey spheres, n = 6) and GBM (brown spheres, n = 8) samples based on 6 differentially expressed proteins (serum amyloid A, hemopexin, apolipoprotein E, haptoglobin, retinol-binding protein and apolipoprotein A-I) identified using DIGE. The axes of the plot indicate PLSDA latent variables t0-t2.](http://www.amritabioquest.org/conference/2013/wp-content/uploads/sites/2/2015/02/sanjeeva-2.jpg)
 Manzoor K, Ph.D.
Manzoor K, Ph.D.
Professor, Centre for Nanoscience & Molecular Medicine, Amrita University
Targeting aberrant cancer kinome using rationally designed nano-polypharmaceutics
Manzoor Koyakutty, Archana Ratnakumary, Parwathy Chandran, Anusha Ashokan, and Shanti Nair
`War on Cancer’ was declared nearly 40 years ago. Since then, we made significant progress on fundamental understanding of cancer and developed novel therapeutics to deal with the most complex disease human race ever faced with. However, even today, cancer remains to be the unconquered `emperor of all maladies’. It is well accepted that meaningful progress in the fight against cancer is possible only with in-depth understanding on the molecular mechanisms that drives its swift and dynamic progression. During the last decade, emerging new technologies such as nanomedicine could offer refreshing life to the `war on cancer’ by way of providing novel methods for molecular diagnosis and therapy.
In the present talk, we discuss our approaches to target critically aberrant cancer kinases using rationally designed polymer-protein and protein-protein core-shell nanomedicines. We have used both genomic and proteomic approaches to identify many intimately cross-linked and complex aberrant protein kinases behind the drug resistance and uncontrolled proliferation of refractory leukemic cells derived from patients. Small molecule inhibitors targeted against oncogenic pathways in these cells were found ineffective due to the involvement of alternative survival pathways. This demands simultaneous inhibition more than one oncogenic kinases using poly-pharmaceutics approach. For this, we have rationally designed core-shell nanomedicines that can deliver several small molecules together for targeting multiple cancer signalling. We have also used combination of small molecules and siRNA for combined gene silencing together with protein kinase inhibition in refractory cancer cells. Optimized nanomedicines were successfully tested in patient samples and found enhanced cytotoxicity and molecular specificity in drug resistant cases.
Nano-polypharmaceutics represents a new generation of nanomedicines that can tackle multiple cancer mechanisms simultaneously. Considering the complexity of the disease, such therapeutic approaches are not simply an advantage, but indispensable.
Acknowledgements:
We thank Dept. of Biotechnology and Dept. Of Science and Technology,Govt. of India for the financial support through `Thematic unit of Excellence in Medical NanoBiotechnology’ and `Nanomedicine- RNAi programs’.

Jaipal Panga Reddy, Dulal Panda and Sanjeeva Srivastava
The rapid emergence of microbial resistance against the ever increasing number of infectious diseases has been a major hurdle in hunt for novel antibiotics and inventive targets required to combat these dreaded pathogens. Despite the discovery of synthetic and semi-synthetic drugs, infectious diseases remain a major cause of concern. The process of drug discovery started from natural products as an antibacterial drug, an old art which was widely adopted in ancient civilizations in India and China. Most of the existing antibiotics are derived from the backbone of natural compounds (Butler M S 2005). Drug discovery process from natural products to synthetic medicine has continued for thousands of years to battle with pathogenic organisms. Although synthetic drugs played a vital role as antimicrobial drugs during the last decade, the over-use of antibiotics has led to the unanticipated changes at the genomic level, resulting into emergence of antibiotic-resistant strains (Singh P et al. 2010). To combat drug-resistant strains there is a need to develop and characterize new drugs by screening natural and synthetic compounds. Curcumin is a well known natural product, with a potential to cure wide range of cancers. Apart from antitumor activity, it also has anti-inflammatory, anti-viral, anti-genotoxic, phototoxic and antimicrobial activities. The exact mechanism of action of curcumin is still obscure and further investigations are required to identify its molecular/cellular targets. Recently, it was observed that curcumin is able to perturb the FtsZ assembly dynamics and elongate the bacterial cell length by inhibiting bacterial cell division (Rai D et al. 2008).
In the present study, we aimed at deciphering the mechanism of action and molecular targets of curcumin in B. subtilis AH75 using classical two dimensional electrophoresis, 2D-difference gel electrophoresis (2DDIGE) in combination with MALDI-TOF/TOFMS. Comparative proteome analysis of control and IC50 curcumin treated B. subtilis has revealed differential expression of 48 proteins (p ≤ 0.05) in response to curcumin treatment. Further, in silico analysis has revealed the involvement of the differential expressed proteins in protein synthesis, transcription/nucleotide synthesis, stress regulation, central metabolism and amino acid metabolic process. Additionally, suppression in major central metabolic dehydrogenase such as glyceraldehyde-3-phosphate dehydrogenase 2, dihydrolipoyl dehydrogenase, malate dehydrogenase, pyruvate dehydrogenase E1 component subunit beta, 2-oxoglutarate dehydrogenase E1 component, ATP synthase epsilon chain, ATP synthase subunit beta and probable NADdependent malic enzyme 1 has been identified (Figure 1). Reduction in expression levels of major dehydrogenases indicates the disturbance in cell membrane integrity to transfer the electrons from NADH to molecular oxygen or maintain the PMF or maintain the membrane potential. Selected potential proteins obtained from proteomic analysis were validated with real-time expression analysis and metabolic assays such as resazurin assay for metabolic activity, CTC assay for respiratory activity and propidium iodide staining for membrane integrity. Findings from this study have revealed that curcumin has potential effects on multiple physiological pathways in bacteria and inhibition of the cell division machinery is one of the prime targets of this drug. Multiple proteins involved in cell division process such as GroEL, ClpC, MetK, Tuf and major dehydrogenases are significantly modulated as a consequence of the drug treatment.





