 Hideaki Nagase, Ph.D.
Hideaki Nagase, Ph.D.
Kennedy Institute of Rheumatology-Centre for Degenerative Diseases, University of Oxford, UK
Osteoarthritis: diagnosis, treatment and challenges
Hideaki Nagase1, Ngee Han Lim1, George Bou-Gharios1, Ernst Meinjohanns2 and Morten Meldal3
- Kennedy Institute of Rheumatology, Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences, University of Oxford, London, W6 8LH UK
- Carlsberg Laboratory, Copenhagen, Denmark,
- Nano-Science Center, Department of Chemistry, University of Copenhagen, Denmark
Osteoarthritis (OA) is the most prevalent age-related degenerative joint disease. With the expanding ageing population, it imposes a major socio-economic burden on society. A key feature of OA is a gradual loss of articular cartilage and deformation of bone, resulting in the impairment of joint function. Currently, there is no effective disease-modifying treatment except joint replacement surgery. There are many possible causes of cartilage loss (e.g. mechanical load, injury, reactive oxygen species, aging, etc.) and etiological factors (obesity, genetics), but the degradation of cartilage is primarily caused by elevated levels of active metalloproteinases. It is therefore attractive to consider proteinase inhibitors as potential therapeutics. However, there are several hurdles to overcome, namely early diagnosis and continuous monitoring of the efficacy of inhibitor therapeutics. We are therefore aiming at developing non-invasive probes to detect cartilage degrading metalloproteinase activities.
We have designed in vivo imaging probes to detect MMP-13 (collagenase 3) activity that participates in OA by degrade cartilage collagen II and MMP-12 (macrophage elastase) activity involved in inflammatory arthritis. These activity-based probes consist of a peptide that is selectively cleaved by the target proteinase, a near-infrared fluorophore and a quencher. The probe’s signal multiplies upon proteolysis. They were first used to follow the respective enzyme activity in vivo in the mouse model of collagen-induced arthritis and we found MMP-12 activity probe (MMP12AP) activation peaked at 5 days after onset of the disease, whereas MMP13AP activation was observed at 10-15 days. The in vivo activation of these probes was inhibited by specific low molecule inhibitors. We proceeded to test both probes in the mouse model of OA induced by the surgical destabilization of medial meniscus of the knee joints. In this model, degradation of knee cartilage is first detected histologically 6 weeks after surgery with significant erosion detectable at 8 weeks. Little activation of MMP12AP was detected, which was expected, as macrophage migration is not obvious in OA. MMP13AP, on the other hand, was significantly activated in the operated knee at 6 weeks compared with the non-operated contralateral knee, but there were no significant differences between the operated and sham-operated knees. At 8 weeks, however, the signals in the operated knees were significantly higher than both the contralateral and sham-operated controls. Activation of aggrecanases and MMP-13 are observed before structural changes of cartilage. We are therefore currently improving the MMP-13 probe for earlier detection by attaching it to polymers that are retained in cartilage.
 Pradip K. Bhatnagar, Ph.D.
Pradip K. Bhatnagar, Ph.D.
Former President & Head, Daiichi Sankyo Life Science Research Centre, India
Strategies for Diseases/Target Selection for Drug Discovery and a Multi-Targeted Approach to Metabolic Disorder
Drug discovery and development is a high risk and expensive undertaking. Although, technologies, such as, bioinformatics, genomics, high throughput screening and computer-aided design have helped identify targets, biomarkers, lead candidates and reduced the time required for advancing an idea from bench to clinic, but it still takes 10-12 years and costs approximately one billion dollars to bring a drug to market globally. Therefore, it is imperative that the strategies to reduce the risk and increase efficiency are carefully selected. In this presentation I would discuss strategies for selecting potential diseases, targets and provide an example of multi-targeted approach to metabolic disorder.
 Sanjeeva Srivastava, Ph.D.
Sanjeeva Srivastava, Ph.D.
Assistant Professor, Proteomics Lab, IIT-Bombay, India
Identification of Potential Early Diagnostic Biomarkers for Gliomas and Various Infectious Diseases using Proteomic Technologies
The spectacular advancements achieved in the field of proteomics research during the last decade have propelled the growth of proteomics for clinical research. Recently, comprehensive proteomic analyses of different biological samples such as serum or plasma, tissue, CSF, urine, saliva etc. have attracted considerable attention for the identification of protein biomarkers as early detection surrogates for diseases (Ray et al., 2011). Biomarkers are biomolecules that can be used for early disease detection, differentiation between closely related diseases with similar clinical manifestations as well as aid in scrutinizing disease progression. Our research group is performing in-depth analysis of alteration in human proteome in different types of brain tumors and various pathogenic infections to obtain mechanistic insight about the disease pathogenesis and host immune responses, and identification of surrogate protein markers for these fatal human diseases.
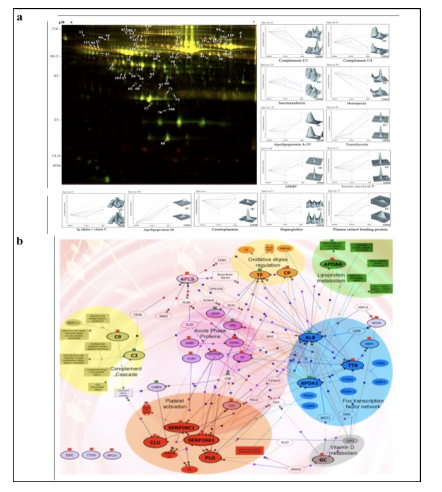
Applying 2D-DIGE in combination with MALDI-TOF/TOF MS we have analyzed the serum and tissue proteome profiles of glioblastoma multiforme; the most common and lethal adult malignant brain tumor (Gollapalli et al., 2012) (Figure 1). Results obtained were validated by employing different immunoassay-based approaches. In serum proteomic analysis we have identified some interesting proteins like haptoglobin, ceruloplasmin, vitamin-D binding protein etc. Moreover, proteomic analysis of different grades (grade-I to IV) of gliomas and normal brain tissue was performed and differential expressions of quite a few proteins such as SIRT2, GFAP, SOD, CDC42 have been identified, which have significant correlation with the tumor growth. While proteomic analysis of cerebrospinal fluid from low grade (grade I & II) vs. high grade (grade III & IV) gliomas revealed modulation of CSF levels of apolipoprotein E, dickkopf related protein 3, vitamin D binding protein and albumin in high grade gliomas. The prospective candidates identified in our studies provide a mechanistic insight of glioma pathogenesis and identification of potential biomarkers. We are also studying the role of JAK/STAT interactome and therapeutic potential of STAT3 inhibitors in gliomas using proteomics approach. Several candidates of the JAK/STAT interactome were identified with altered expression and a significant correlation was observed between STAT3 and PDK1 transcript expression level.
We have also investigated the changes in human serum proteome in different infectious diseases including falciparum and vivax malaria (Ray et al., 2012a; Ray et al., 2012b), dengue (Ray et al., 2012c) and leptospirosis (Srivastava et al., 2012). Although, quite a few serum proteins were found to be commonly altered in different infectious diseases and might be a consequence of inflammation mediated acute phase response signaling, uniquely modulated candidates were identified in each pathogenic infection indicating the some inimitable responses. Further, a panel of identified proteins consists of six candidates; serum amyloid A, hemopexin, apolipoprotein E, haptoglobin, retinol-binding protein and apolipoprotein A-I was used to build statistical sample class prediction models employing PLSDA and other classification methods to predict the clinical phenotypic classes and 91.37% overall prediction accuracy was achieved (Figure 2). ROC curve analysis was carried out to evaluate the individual performance of classifier proteins. The excellent discrimination among the different disease groups on the basis of differentially expressed proteins demonstrates the potential diagnostic implications of this analytical approach.
Keywords: Diagnostic biomarkers, Gliomas, Infectious Diseases, Proteomics, Serum proteome
Acknowledgments: This disease biomarker discovery research was supported by Department of Biotechnology, India grant (No. BT/PR14359/MED/30/916/2010), Board of Research in Nuclear Sciences (BRNS) DAE young scientist award (2009/20/37/4/BRNS) and a startup grant 09IRCC007 from the IIT Bombay. The active support from Advanced Center for Treatment Research and Education in Cancer (ACTREC), Tata Memorial Hospital (TMH), and Seth GS Medical College and KEM Hospital Mumbai, India in clinical sample collection process is gratefully acknowledged.
References :
- Ray S, Reddy PJ, Jain R, Gollapalli K. Moiyadi A, Srivastava S. Proteomic technologies for the identification of disease biomarkers in serum: advances and challenges ahead. Proteomics 11: 2139-61, 2011.
- Gollapalli K, Ray S, Srivastava R, Renu D, Singh P, Dhali S, Dikshit JB, Srikanth R, Moiyadi A, Srivastava S. Investigation of serum proteome alterations in human glioblastoma multiforme. Proteomics 12(14): 2378-90, 2012.
- Ray S, Renu D, Srivastava R, Gollapalli K, Taur S, Jhaveri T, Dhali S, Chennareddy S, Potla A, Dikshit JB, Srikanth R, Gogtay N, Thatte U, Patankar S, Srivastava S. Proteomic investigation of falciparum and vivax malaria for identification of surrogate protein markers. PLoS One 7(8): e41751, 2012a.
- Ray S, Kamath KS, Srivastava R, Raghu D, Gollapalli K, Jain R, Gupta SV, Ray S, Taur S, Dhali S, Gogtay N, Thatte U, Srikanth R, Patankar S, Srivastava S. Serum proteome analysis of vivax malaria: An insight into the disease pathogenesis and host immune response. J Proteomics 75(10): 3063-80, 2012b.
- Srivastava R, Ray S, Vaibhav V, Gollapalli K, Jhaveri T, Taur S, Dhali S, Gogtay N, Thatte U, Srikanth R, Srivastava S. Serum profiling of leptospirosis patients to investigate proteomic alterations. J Proteomics 76: 56-68, 2012.
- Ray S, Srivastava R, Tripathi K, Vaibhav V, Srivastava S. Serum proteome changes in dengue virus-infected patients from a dengue-endemic area of India: towards new molecular targets? OMICS 16(10): 527-36, 2012c.
* Correspondence: Dr. Sanjeeva Srivastava, Department of Biosciences and Bioengineering, IIT Bombay, Mumbai 400 076, India: E-mail: sanjeeva@iitb.ac.in; Phone: +91-22-2576-7779, Fax: +91-22-2572-3480
![Figure 2 (a) Western blot analysis of haptoglobin (HP), serum amyloid A (SAA), and clusterin (CLU) from serum samples of healthy control (HC) [n = 12], falciparum malaria (FM) [n = 12], vivax malaria (VM) [n = 12], Leptospirosis (Lep) [n = 6], dengue fever [DF] [n = 6] and non infectious disease control (NIDC:GBM) [n = 12]. Representative blots of the target proteins are depicted along with their respective relative abundance volumes (volume X 104). All the data are represented as mean ± SE. (b) Discrimination of malaria from dengue, leptospirosis and GBM using PLS-DA analysis. PLS-DA scores Plot for FM (blue spheres, n = 8), VM (green spheres, n = 8), DF (red spheres, n = 6), Lep (grey spheres, n = 6) and GBM (brown spheres, n = 8) samples based on 6 differentially expressed proteins (serum amyloid A, hemopexin, apolipoprotein E, haptoglobin, retinol-binding protein and apolipoprotein A-I) identified using DIGE. The axes of the plot indicate PLSDA latent variables t0-t2.](http://www.amritabioquest.org/conference/2013/wp-content/uploads/sites/2/2015/02/sanjeeva-2.jpg)
 Kal Ramnarayan, Ph.D.
Kal Ramnarayan, Ph.D.
Co-founder President & Chief Scientific Officer, Sapient Discovery, San Diego, CA, USA
A cost-effective approach to Protein Structure-guided Drug Discovery: Aided by Bioinformatics, Chemoinformatics and computational chemistry
With the mapping of the human genome completed almost a decade ago, efforts are still underway to understand the gene products (i.e., proteins) in the human biological and disease pathways. Deciphering such information is very important for the discovery and development of small molecule drugs as well as protein therapeutics for various human diseases for which no cure exists. As an example, with more than 500 members, the kinase family of protein targets continues to be an important and attractive class for drug discovery. While how many of the members in this family are actually druggable is still to be established, there are several ongoing efforts on this class of proteins across a broad spectrum of disease categories. Even though in general the protein structural topology might looks similar, there are issues with respect selectivity of identified small molecule inhibitors when, the lead molecule discovery is carried out at the ATP binding site. As an added complexity, allosteric modulators are needed for some of the members, but the actual site for such modulation on the protein target can not resolved with uncertainty. In this presentation we will describe a bioinformatics and computational based platform for small molecule discovery for protein targets that are involved in protein-protein interactions as well as targets like kinases and phosphatases. We will describe a computational approach in which we have used an informatics based platform with several hundred kinases to sort through in silico and identify inhibitors that are likely to be highly selective in the lead generation phase. We will discuss the implication of this approach on the drug discovery of the kinase and phosphatase classes in general and independent of the disease category.

Jaipal Panga Reddy, Dulal Panda and Sanjeeva Srivastava
The rapid emergence of microbial resistance against the ever increasing number of infectious diseases has been a major hurdle in hunt for novel antibiotics and inventive targets required to combat these dreaded pathogens. Despite the discovery of synthetic and semi-synthetic drugs, infectious diseases remain a major cause of concern. The process of drug discovery started from natural products as an antibacterial drug, an old art which was widely adopted in ancient civilizations in India and China. Most of the existing antibiotics are derived from the backbone of natural compounds (Butler M S 2005). Drug discovery process from natural products to synthetic medicine has continued for thousands of years to battle with pathogenic organisms. Although synthetic drugs played a vital role as antimicrobial drugs during the last decade, the over-use of antibiotics has led to the unanticipated changes at the genomic level, resulting into emergence of antibiotic-resistant strains (Singh P et al. 2010). To combat drug-resistant strains there is a need to develop and characterize new drugs by screening natural and synthetic compounds. Curcumin is a well known natural product, with a potential to cure wide range of cancers. Apart from antitumor activity, it also has anti-inflammatory, anti-viral, anti-genotoxic, phototoxic and antimicrobial activities. The exact mechanism of action of curcumin is still obscure and further investigations are required to identify its molecular/cellular targets. Recently, it was observed that curcumin is able to perturb the FtsZ assembly dynamics and elongate the bacterial cell length by inhibiting bacterial cell division (Rai D et al. 2008).
In the present study, we aimed at deciphering the mechanism of action and molecular targets of curcumin in B. subtilis AH75 using classical two dimensional electrophoresis, 2D-difference gel electrophoresis (2DDIGE) in combination with MALDI-TOF/TOFMS. Comparative proteome analysis of control and IC50 curcumin treated B. subtilis has revealed differential expression of 48 proteins (p ≤ 0.05) in response to curcumin treatment. Further, in silico analysis has revealed the involvement of the differential expressed proteins in protein synthesis, transcription/nucleotide synthesis, stress regulation, central metabolism and amino acid metabolic process. Additionally, suppression in major central metabolic dehydrogenase such as glyceraldehyde-3-phosphate dehydrogenase 2, dihydrolipoyl dehydrogenase, malate dehydrogenase, pyruvate dehydrogenase E1 component subunit beta, 2-oxoglutarate dehydrogenase E1 component, ATP synthase epsilon chain, ATP synthase subunit beta and probable NADdependent malic enzyme 1 has been identified (Figure 1). Reduction in expression levels of major dehydrogenases indicates the disturbance in cell membrane integrity to transfer the electrons from NADH to molecular oxygen or maintain the PMF or maintain the membrane potential. Selected potential proteins obtained from proteomic analysis were validated with real-time expression analysis and metabolic assays such as resazurin assay for metabolic activity, CTC assay for respiratory activity and propidium iodide staining for membrane integrity. Findings from this study have revealed that curcumin has potential effects on multiple physiological pathways in bacteria and inhibition of the cell division machinery is one of the prime targets of this drug. Multiple proteins involved in cell division process such as GroEL, ClpC, MetK, Tuf and major dehydrogenases are significantly modulated as a consequence of the drug treatment.
The global healthcare scene of which the pharmaceutical industry and its products are integral components is today at the cross roads. The high and unaffordable costs of drug research with estimates of over 1 billion dollars for every new drug discovered and developed, the very low success rates, the high degree of obsolescence due to undesirable adverse drug reactions, the decline in the development pipeline of new drugs, patent expiries leading to generic competition and the public’s disillusionment with use of chemicals for human consumption as drugs have all significantly contributed to the problems of this lifeline industry. The strategy adopted by the large R&D based Corporations to get bigger and bigger through mergers and acquisitions to improve cost-effectiveness and productivity of R&D has so far not worked effectively. Consequently, one of the recent trends in healthcare, articulated by many experts is to look for alternate or even complementary approaches to reduce the impact of rising costs of drugs on healthcare. Various new strategies for drug discovery such as the use of Natural Products especially medicinal plants are being actively pursued by healthcare planners and providers. Side by side, traditional systems of medicine whether from the oriental countries or the western nations are also having a serious relook to understand their usefulness in healthcare. To achieve its legitimate position in the healthcare scenario, it is essential to scientifically validate their claimed utility through appropriate and systematic research efforts including pre-clinical and clinical studies. In addition to their own use as medicines, knowledge on the Indian Traditional Medicines can be used as a platform for new drug discovery. The huge potential for carrying out systematic R&D programs for new Drug Discovery based on natural products and possible strategies to realise them in the coming decades will be explained in this presentation.
 Akhilesh Pandey, Ph.D.
Akhilesh Pandey, Ph.D.
Professor, Johns Hopkins University School of Medicine, Baltimore, USA
A draft map of the human proteome
We have generated a draft map of the human proteome through a systematic and comprehensive analysis of normal human adult tissues, fetal tissues and hematopoietic cells as an India-US initiative. This unique dataset was generated from 30 histologically normal adult tissues, fetal tissues and purified primary hematopoietic cells that were analyzed at high resolution in the MS mode and by HCD fragmentation in the MS/MS mode on LTQ-Orbitrap Velos/Elite mass spectrometers. This dataset was searched against a 6-frame translation of the human genome and RNA-Seq transcripts in addition to standard protein databases. In addition to confirming a large majority (>16,000) of the annotated protein-coding genes in humans, we obtained novel information at multiple levels: novel protein-coding genes, unannotated exons, novel splice sites, proof of translation of pseudogenes (i.e. genes incorrectly annotated as pseudogenes), fused genes, SNPs encoded in proteins and novel N-termini to name a few. Many proteins identified in this study were identified by proteomic methods for the first time (e.g. hypothetical proteins or proteins annotated based solely on their chromosomal location). We have generated a catalog of proteins that show a more tissue-restricted pattern of expression, which should serve as the basis for pursuing biomarkers for diseases pertaining to specific organs. This study also provides one of the largest sets of proteotypic peptides for use in developing MRM assays for human proteins. Identification of several novel protein-coding regions in the human genome underscores the importance of systematic characterization of the human proteome and accurate annotation of protein-coding genes. This comprehensive dataset will complement other global HUPO initiatives using antibody-based as well as MRM mass spectrometry-based strategies. Finally, we believe that this dataset will become a reference set for use as a spectral library as well as for interesting interrogations pertaining to biomedical as well as bioinformatics questions.
 Ramani A. Aiyer, Ph.D., MBA
Ramani A. Aiyer, Ph.D., MBA
Principal, Shasta BioVentures, San Jose, CA, USA
New Drug R&D in India: Challenges & Opportunities
New drug discovery and development has become a global endeavor, with Western big pharmaceutical companies farming out more and more chemistry and biology research to Asia, particularly India and China. During the last decade, several Indian pharmaceutical companies have embarked on ambitious R&D programs, with slow but steady progress in developing new chemical / molecular entities. The Indian government has also made a strong commitment to promote innovation and entrepreneurship in the biotechnology sector. The first part of the talk will focus on a case study showing the entire process of discovery and development of a new drug recently launched for Rheumatoid Arthritis. We will then address the challenges of conducting innovative R&D in India and actions necessary to overcome them. The second part of the talk will make the case for developing Ayurvedic drug formulations for the Western / Global markets, again using the example of Rheumatoid Arthritis (Aamavaata). Ayurveda takes a holistic approach to disease diagnosis and therapy based on interactions among body type (prakriti), tri-doshas (three body humors), sapta-dhatus (seven tissues) and malas (excretions). The drugs prescribed are usually herbo-mineral formulations comprising multiple medicinal plants and / or metals. The manufacturing processes date back to Ayurvedic texts several thousand years old, and are compiled in the Ayurvedic Pharmacopeia. Also, the treatment modalities and drug formulations are “personalized” to fit different patient types, based on the holistic diagnoses mentioned earlier. There is a tremendous need to establish a sound basis for Ayurvedic drug discovery R&D for the modern world. We must find a scientific and ethical way to leverage the vast body of anecdotal and possibly retrospective data on patients undergoing Ayurvedic treatment. Combined with in vitro and in vivo biological data on Ayurvedic herbo-mineral formulations, the adoption of stringent manufacturing practices, and designing sound clinical trials to establish the safety and efficacy, India has a golden opportunity to expand the reach of Ayurvedic drugs into Western / Global medical practice.

Syed Salman Lateef and Vinayak A K
Development of Supercritical Fluid Chromatography methods for the replacement of existing USP Normal phase liquid chromatography methods
Normal phase liquid chromatography methods often have long run times and involve environmentally toxic/costly solvents. Supercritical chromatography methods on the other hand are faster, inexpensive, and eco-friendly. The low viscous supercritical carbon dioxide operates at high flow rates compared to LC without losing separation efficiency. In this work, SFC methods are developed to replace three United States Pharmacopeial (USP) normal phase achiral methods – prednisolone, tolazamide and cholecalciferol. System suitability parameters of the normal phase method are compared against the SFC method. Precision, linearity and robustness of the new SFC methods are demonstrated. SFC methods were found to be cost effective in terms of analysis time and solvent savings. The SFC method does not require purchase and disposal of expensive environmentally hazardous chemicals. Hence, the newly developed SFC method provides a faster and safer solution.
 Sudarslal S, Ph.D.
Sudarslal S, Ph.D.
Associate Professor, School of Biotechnology, Amrita University
Electrospray ionization ion trap mass spectrometry for cyclic peptide characterization
There has been considerable interest in the isolation and structural characterization of bioactive peptides produced by bacteria and fungi. Most of the peptides are cyclic depsipeptides characterized by the presence of lactone linkages and β-hydroxy fatty acids. Occurrence of microheterogeneity is another remarkable property of these peptides. Even if tandem mass spectrometers are good analytical tools to structurally characterize peptides and proteins, sequence analysis of cyclic peptides is often ambiguous due to the random ring opening of the peptides and subsequent generation of a set of linear precursor ions with the same m/z. Here we report combined use of chemical derivatization and multistage fragmentation capability of ion trap mass spectrometers to determine primary sequences of a series of closely related cyclic peptides.

Ravindra Gudihal, Suresh Babu C V
Bioanalytical Characterization of Therapeutic Proteins
The characterization of therapeutic proteins such as monoclonal antibody (mAb) during different stages of manufacturing is crucial for timely and successful product release. Regulatory agencies require a variety of analytical technologies for comprehensive and efficient protein analysis. Electrophoresis-based techniques and liquid chromatography (LC) either standalone or coupled to mass spectrometry (MS) are at the forefront for the in-depth analysis of protein purity, isoforms, stability, aggregation, posttranslational modifications, PEGylation, etc. In this presentation, a combination of various chromatographic and electrophoretic techniques such as liquid-phase isoelectric focusing, microfluidic and capillary-based electrophoresis (CE), liquid chromatography (LC) and combinations of those with mass spectrometry techniques will be discussed. We present a workflow based approach to the analysis of therapeutic proteins. In successive steps critical parameters like purity, accurate mass, aggregation, peptide sequence, glycopeptide and glycan analysis are analyzed. In brief, the workflow involved proteolytic digestion of therapeutic protein for peptide mapping, N-Glycanase and chemical labeling reaction for glycan analysis, liquid-phase isoelectric focusing for enrichment of charge variants followed by a very detailed analysis using state of the art methods such as CE-MS and LC-MS. For the analysis of glycans, we use combinations of CE-MS and LC-MS to highlight the sweet spots of these techniques. CE-MS is found to be more useful in analysis of highly sialylated glycans (charged glycans) while nano LC-MS seems to be better adapted for analysis of neutral glycans. These two techniques can be used to get complementary data to profile all the glycans present in a given protein. In addition, microfluidic electrophoresis was used as a QC tool in initial screening for product purity, analysis of papain digestion fragments of mAb, protein PEGylation products, etc. The described workflow involves multiple platforms, provides an end to end solution for comprehensive protein characterization and aims at reducing the total product development time.

Tejaswini Subbannayya, Nandini A. Sahasrabuddhe, Arivusudar Marimuthu, Santosh Renuse, Gajanan Sathe, Srinivas M. Srikanth, Mustafa A. Barbhuiya, Bipin Nair, Juan Carlos Roa, Rafael Guerrero-Preston, H. C. Harsha, David Sidransky, Akhilesh Pandey, T. S. Keshava Prasad and Aditi Chatterjee
Proteomic profiling of gallbladder cancer secretome – a source for circulatory biomarker discovery
Gallbladder cancer (GBC) is the fifth most common cancer of the gastrointestinal tract and one of the common malignancies that occur in the biliary tract (Misra et al. 2006; Lazcano-Ponce et al. 2001). It has a poor prognosis with survival of less than 5 years in 90% of the cases (Misra et al. 2003). The etiology is ill-defined. Several risk factors have been reported including cholelithiasis, obesity, female gender and exposure to carcinogens (Eslick 2010; Kumar et al. 2006). Poor prognosis in GBC is mainly due to late presentation of the disease and lack of reliable biomarkers for early diagnosis. This emphasizes the need to identify and characterize cancer biomarkers to aid in the diagnosis and prognosis of GBC. Secreted proteins are an important class of molecules which can be detected in body fluids and has been targeted for biomarker discovery. There are challenges faced in the proteomic interrogation of body fluids especially plasma such as low abundance of tumor secreted proteins, high complexity and high abundance of other proteins that are not released by the tumor cells (Tonack et al. 2009). Profiling of conditioned media from the cancer cell lines can be used as an alternate means to identify secreted proteins from tumor cells (Kashyap et al. 2010; Marimuthu et al. 2012). We analyzed the invasive property of 7 GBC cell lines (SNU-308, G-415, GB-d1, TGBC2TKB, TGBC24TKB, OCUG-1 and NOZ). Four cell lines were selected for analysis of the cancer secretome based on the invasive property of the cells. We employed isobaric tags for relative and absolute quantitation (iTRAQ) labeling technology coupled with high resolution mass spectrometry to identify and characterize secretome from the panel of 4GBC cancer cells mentioned above. In total, we have identified around 2,000 proteins of which 175 were secreted at differential abundance across all the four cell lines. This secretome analysis will act as a reservoir of candidate biomarkers. Currently, we are investigating and validating these candidate markers from GBC cell secretome. Through this study, we have shown mass spectrometry-based quantitative proteomic analysis as a robust approach to investigate secreted proteins in cancer cells.










