 Sanjeeva Srivastava, Ph.D.
Sanjeeva Srivastava, Ph.D.
Assistant Professor, Proteomics Lab, IIT-Bombay, India
Identification of Potential Early Diagnostic Biomarkers for Gliomas and Various Infectious Diseases using Proteomic Technologies
The spectacular advancements achieved in the field of proteomics research during the last decade have propelled the growth of proteomics for clinical research. Recently, comprehensive proteomic analyses of different biological samples such as serum or plasma, tissue, CSF, urine, saliva etc. have attracted considerable attention for the identification of protein biomarkers as early detection surrogates for diseases (Ray et al., 2011). Biomarkers are biomolecules that can be used for early disease detection, differentiation between closely related diseases with similar clinical manifestations as well as aid in scrutinizing disease progression. Our research group is performing in-depth analysis of alteration in human proteome in different types of brain tumors and various pathogenic infections to obtain mechanistic insight about the disease pathogenesis and host immune responses, and identification of surrogate protein markers for these fatal human diseases.
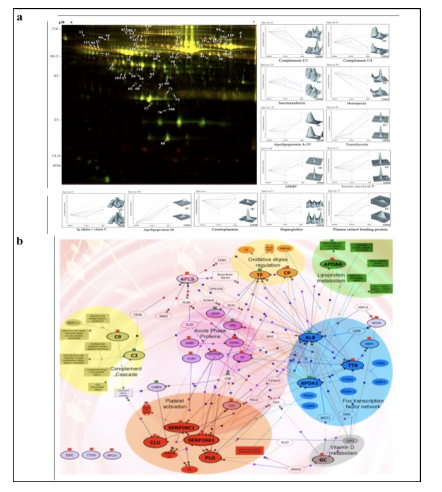
Applying 2D-DIGE in combination with MALDI-TOF/TOF MS we have analyzed the serum and tissue proteome profiles of glioblastoma multiforme; the most common and lethal adult malignant brain tumor (Gollapalli et al., 2012) (Figure 1). Results obtained were validated by employing different immunoassay-based approaches. In serum proteomic analysis we have identified some interesting proteins like haptoglobin, ceruloplasmin, vitamin-D binding protein etc. Moreover, proteomic analysis of different grades (grade-I to IV) of gliomas and normal brain tissue was performed and differential expressions of quite a few proteins such as SIRT2, GFAP, SOD, CDC42 have been identified, which have significant correlation with the tumor growth. While proteomic analysis of cerebrospinal fluid from low grade (grade I & II) vs. high grade (grade III & IV) gliomas revealed modulation of CSF levels of apolipoprotein E, dickkopf related protein 3, vitamin D binding protein and albumin in high grade gliomas. The prospective candidates identified in our studies provide a mechanistic insight of glioma pathogenesis and identification of potential biomarkers. We are also studying the role of JAK/STAT interactome and therapeutic potential of STAT3 inhibitors in gliomas using proteomics approach. Several candidates of the JAK/STAT interactome were identified with altered expression and a significant correlation was observed between STAT3 and PDK1 transcript expression level.
We have also investigated the changes in human serum proteome in different infectious diseases including falciparum and vivax malaria (Ray et al., 2012a; Ray et al., 2012b), dengue (Ray et al., 2012c) and leptospirosis (Srivastava et al., 2012). Although, quite a few serum proteins were found to be commonly altered in different infectious diseases and might be a consequence of inflammation mediated acute phase response signaling, uniquely modulated candidates were identified in each pathogenic infection indicating the some inimitable responses. Further, a panel of identified proteins consists of six candidates; serum amyloid A, hemopexin, apolipoprotein E, haptoglobin, retinol-binding protein and apolipoprotein A-I was used to build statistical sample class prediction models employing PLSDA and other classification methods to predict the clinical phenotypic classes and 91.37% overall prediction accuracy was achieved (Figure 2). ROC curve analysis was carried out to evaluate the individual performance of classifier proteins. The excellent discrimination among the different disease groups on the basis of differentially expressed proteins demonstrates the potential diagnostic implications of this analytical approach.
Keywords: Diagnostic biomarkers, Gliomas, Infectious Diseases, Proteomics, Serum proteome
Acknowledgments: This disease biomarker discovery research was supported by Department of Biotechnology, India grant (No. BT/PR14359/MED/30/916/2010), Board of Research in Nuclear Sciences (BRNS) DAE young scientist award (2009/20/37/4/BRNS) and a startup grant 09IRCC007 from the IIT Bombay. The active support from Advanced Center for Treatment Research and Education in Cancer (ACTREC), Tata Memorial Hospital (TMH), and Seth GS Medical College and KEM Hospital Mumbai, India in clinical sample collection process is gratefully acknowledged.
References :
- Ray S, Reddy PJ, Jain R, Gollapalli K. Moiyadi A, Srivastava S. Proteomic technologies for the identification of disease biomarkers in serum: advances and challenges ahead. Proteomics 11: 2139-61, 2011.
- Gollapalli K, Ray S, Srivastava R, Renu D, Singh P, Dhali S, Dikshit JB, Srikanth R, Moiyadi A, Srivastava S. Investigation of serum proteome alterations in human glioblastoma multiforme. Proteomics 12(14): 2378-90, 2012.
- Ray S, Renu D, Srivastava R, Gollapalli K, Taur S, Jhaveri T, Dhali S, Chennareddy S, Potla A, Dikshit JB, Srikanth R, Gogtay N, Thatte U, Patankar S, Srivastava S. Proteomic investigation of falciparum and vivax malaria for identification of surrogate protein markers. PLoS One 7(8): e41751, 2012a.
- Ray S, Kamath KS, Srivastava R, Raghu D, Gollapalli K, Jain R, Gupta SV, Ray S, Taur S, Dhali S, Gogtay N, Thatte U, Srikanth R, Patankar S, Srivastava S. Serum proteome analysis of vivax malaria: An insight into the disease pathogenesis and host immune response. J Proteomics 75(10): 3063-80, 2012b.
- Srivastava R, Ray S, Vaibhav V, Gollapalli K, Jhaveri T, Taur S, Dhali S, Gogtay N, Thatte U, Srikanth R, Srivastava S. Serum profiling of leptospirosis patients to investigate proteomic alterations. J Proteomics 76: 56-68, 2012.
- Ray S, Srivastava R, Tripathi K, Vaibhav V, Srivastava S. Serum proteome changes in dengue virus-infected patients from a dengue-endemic area of India: towards new molecular targets? OMICS 16(10): 527-36, 2012c.
* Correspondence: Dr. Sanjeeva Srivastava, Department of Biosciences and Bioengineering, IIT Bombay, Mumbai 400 076, India: E-mail: sanjeeva@iitb.ac.in; Phone: +91-22-2576-7779, Fax: +91-22-2572-3480
![Figure 2 (a) Western blot analysis of haptoglobin (HP), serum amyloid A (SAA), and clusterin (CLU) from serum samples of healthy control (HC) [n = 12], falciparum malaria (FM) [n = 12], vivax malaria (VM) [n = 12], Leptospirosis (Lep) [n = 6], dengue fever [DF] [n = 6] and non infectious disease control (NIDC:GBM) [n = 12]. Representative blots of the target proteins are depicted along with their respective relative abundance volumes (volume X 104). All the data are represented as mean ± SE. (b) Discrimination of malaria from dengue, leptospirosis and GBM using PLS-DA analysis. PLS-DA scores Plot for FM (blue spheres, n = 8), VM (green spheres, n = 8), DF (red spheres, n = 6), Lep (grey spheres, n = 6) and GBM (brown spheres, n = 8) samples based on 6 differentially expressed proteins (serum amyloid A, hemopexin, apolipoprotein E, haptoglobin, retinol-binding protein and apolipoprotein A-I) identified using DIGE. The axes of the plot indicate PLSDA latent variables t0-t2.](http://www.amritabioquest.org/conference/2013/wp-content/uploads/sites/2/2015/02/sanjeeva-2.jpg)
 Leland H. Hartwell Ph.D.
Leland H. Hartwell Ph.D.
2001 Nobel Laureate, Physiology & Medicine
Dr. Lee Hartwell received the 2001 Nobel Prize in Physiology / Medicine for his discovery of protein molecules that control the division of cells. He was the President and Director of the Fred Hutchinson Cancer Research Center in Seattle, Washington before moving to Arizona State University’s Center for Sustainable Health.
Dr. Hartwell is also adjunct faculty at Amrita University. He spoke to the delegates at Bioquest from his office in the US, over Amrita’s e-learning platform A-View. Given below are excerpts from his address.
I would like to address the young people in the audience. I know that many of you may have come to this meeting wondering, “How can I become a successful scientist? How can I prepare myself to make a contribution in this world?”
These questions are interesting to me also.
Believe it or not, I am still trying to be a successful scientist. That may surprise you since you probably think that a Nobel laureate must have found the answers. But the problem is that the answers to these questions change with time and the answers are different today than what they were when I began my career fifty years ago. The strategy of the 1960’s doesn’t work so well anymore. What is different now?
First, what we know now is much more. For example, by 1970, no genes from any organisms were sequenced. In 2013, we have the complete sequence of the human genome. Second, not only do we know much more today, accessing that knowledge is easy. Third, obtaining new information is much faster today.
Our rich understanding of science and technology is now needed to solve many serious problems. The human population has reached the size where we are utilizing all available resource of the planet. We are utilizing all of the agricultural land, all of the water, all of the forest and fishing resources. We are also polluting the planet that we live on.
We are polluting the land with fertilizers and pesticides; the oceans with acids and the atmosphere with carbon dioxide. We are using up top soil and ground water, thereby reducing our capacity to feed ourselves. We are using up petroleum, the energy source that our entire economy is dependent on. These are problems we were largely unaware of, fifty years ago. But these are problems that must be solved in your life times.
The big question facing your generation is, how can human beings live sustainably on planet earth. Your two broad goals on sustainability are 1) leave the planet as you first found it for your future generations; don’t use up the resources and don’t pollute the planet 2) everyone deserves to have an equal share of the earth’s resources.
Income strongly determines one’s opportunities in life. Many poor people succumb to chronic diseases and unhealthy environments. This inequality undermines our ability to live sustainably. We can’t ask the poor to leave the planet as they found it if they can’t support their families. Education, healthcare, employment are essential to having a sustainable society.
How can we be a successful scientist in 2013?
1. First choose a problem to solve
2. Ask questions to understand why it is not solved
3. Collaborate with those who can help
4. Develop a solution that works in the real worldChronic diseases are our major burden and this burden will get worse. Heart disease, diabetes, cancer, dementia and other diseases. The good news is that the chronic diseases are largely preventable and more easily curable if detected early. One question that attracts me is how can we detect disease earlier when it can be more easily cured?
Can we use our increasing knowledge in molecular biology to identify biomarkers for early disease detection?
We need to collaborate very closely with clinicians who care for patients to find out exactly where they need help.
I think if we apply our technology to important clinical questions we will actually save medical expenditure and be well on our way to making a great contribution to society.
 Michelle Hermiston, MD, Ph.D.
Michelle Hermiston, MD, Ph.D.
Assistant Professor, Department of Pediatrics University of California San Francisco, USA
Interrogating Signaling Networks at the Single Cell Level In Primary Human Patient Samples
Multiparameter phosphoflow cytometry is a highly sensitive proteomic approach that enables monitoring of biochemical perturbations at the single cell level. By combining antisera to cell surface markers and key intracellular proteins, perturbations in signaling networks, cell survival and apoptosis mediators, cell cycle regulators, and/or modulators of other cellular processes can be analyzed in a highly reproducible and sensitive manner in the basal state and in response to stimulation or drug treatment. Advantages of this approach include the ability to identify the biochemical consequences of genetic and/or epigenetic changes in small numbers of cells, to map potential interplay between various signaling networks simultaneously in a single cell, and to interrogate potential mechanisms of drug resistance or response in a primary patient sample. Application of this technology to patients with acute lymphoblastic leukemia or the autoimmune disease systemic lupus erythematosus (SLE) will be discussed.
 Lalitha Subramanian, Ph.D.
Lalitha Subramanian, Ph.D.
Chief Scientific Officer & VP, Services at Scienomics, USA
Nanoscale Simulations – Tackling Form and Formulation Challenges in Drug Development and Drug Delivery
Lalitha Subramanian, Dora Spyriouni, Andreas Bick, Sabine Schweizer, and Xenophon Krokidis Scienomics
The discovery of a compound which is potent in activity against a target is a major milestone in Pharmaceutical and Biotech industry. However, a potent compound is only effective as a therapeutic agent when it can be administered such that the optimal quantity is transported to the site of action at an optimal rate. The active pharmaceutical ingredient (API) has to be tested for its physicochemical properties before the appropriate dosage form and formulation can be designed. Some of the commonly evaluated parameters are crystal forms and polymorphs, solubility, dissolution behavior, stability, partition coefficient, water sorption behavior, surface properties, particle size and shape, etc. Pharmaceutical development teams face the challenge of quickly and efficiently determining a number of properties with small quantities of the expensive candidate compounds. Recently the trend has been to screen these properties as early as possible and often the candidate compounds are not available in sufficient quantities. Increasingly, these teams are leveraging nanoscale simulations similar to those employed by drug discovery teams for several decades. Nanoscale simulations are used to predict the behavior using very little experimental data and only if this is promising further experiments are done. Another aspect where nanoscale simulations are being used in drug development and drug delivery is to get insights into the behavior of the system so that process failures can be remediated and formulation performance can be improved. Thus, the predictive screening and the in-depth understanding leads to experimental efficiency resulting in far-reaching business impacts.
With specific examples, this talk will focus on the different types of nanoscale simulations used to predict properties of the API in excipients and also provide insight into system behavior as a function of shelf life, temperature, mechanical stress, etc.

Pandiaraj Manickam, Niroj Kumar Sethy, Kalpana Bhargava, Vepa Kameswararao and Karunakaran Chandran
Designing electrochemical label free immunosensors for cytochrome c using nanocomposites functionalized screen printed electrodes
Release of cytochrome c (cyt c) from mitochondria into cytosol is a hallmark of apoptosis, used as a biomarker of mitochondrial dependent pathway of cell death (Kluck et al. 1997; Green et al. 1998). We have previously reported cytochrome c reductase (CcR) based biosensors for the measurement of mitochondrial cyt c release (Pandiaraj et al. 2013). Here, we describe the development of novel label-free, immunosensor for cyt c utilizing its specific monoclonal antibody. Two types of nanocomposite modified immunosensing platforms were used for the immobilization of anti-cyt c; (i) Self-assembled monolayer (SAM) functionalized gold nanoparticles (GNP) in conducting polypyrrole (PPy) modified screen printed electrodes (SPE) (ii) Carbon nanotubes (CNT) incorporated PPy on SPE. The nanotopologies of the modified electrodes were confirmed by scanning electron microscopy (SEM). Cyclic voltammetry, electrochemical impedance spectroscopy (EIS) were used for probing the electrochemical properties of the nanocomposite modified electrodes. Method for cyt c quantification is based on the direct electron transfer between Fe3+/Fe2+-heme of cyt c selectively bound to anti-cyt c modified electrode. The Faradaic current response of these nanoimmunosensor increases with increase in cyt c concentration. The procedure for cyt c detection was also optimized (pH, incubation times, and characteristics of electrodes) to improve the analytical characteristics of immunosensors. The analytical performance of anti-cyt c biofunctionalized GNP-PPy nanocomposite platform (detection limit 0.5 nM; linear range: 0.5 nM–2 μM) was better than the CNT-PPy (detection limit 2 nM; linear range: 2 nM-500nM). The detection limits were well below the normal physiological concentration range (Karunakaran et al. 2008). The proposed method does not require any signal amplification or labeled secondary antibodies contrast to widespread ELISA and Western blot. The immunosensors results in simple and rapid measurement of cyt c and has great potential to become an inexpensive and portable device for conventional clinical immunoassays.

John Stanley, Satheesh Babu, Ramacahandran T and Bipin Nair
Pt-Pd decorated TiO2 nanotube array for the non-enzymatic determination of glucose in neutral medium
Rapidly expanding diabetic population and the complications associated with elevated glycemic levels necessitates the need for a highly sensitive, selective and stable blood glucose measurement strategy. The high sensitivity and selectivity of enzymatic sensors together with viable manufacturing technologies such as screen-printing have made a great social and economic impact. However, the intrinsic nature of the enzymes leads to lack of stability and consequently reduces shelf life and imposes the need for stringent storage conditions. As a result much effort has been directed towards the development of ‘enzyme-free’ glucose sensors (Park et al. 2006). In this paper, a non-enzymatic amperometric sensor for selective and sensitive direct electrooxidation of glucose in neutral medium was fabricated based on Platinum-Palladium (Pt–Pd) nanoparticle decorated titanium dioxide (TiO2) nanotube arrays. Highly ordered TiO2 nanotube arrays were obtained using a single step anodization process (Grimes C A and Mor G K 2009) over which Pt–Pd nanoparticles where electrochemically deposited. Scanning Electron Microscopy (SEM) analysis revealed the diameter of the TiO2 nanotubes to be approximately 40 nm. Elemental analysis after electrochemical deposition confirms the presence of Pt–Pd. Electrochemical characterization of the sensor was carried out using cyclic voltammetry technique (−1.0 to +1.0V) in phosphate buffer saline (PBS) pH 7.4. All further glucose oxidation studies were performed in PBS (pH 7.4). The sensor exhibited good linear response towards glucose for a concentration range of 1 μM to 20mM with a linear regression coefficient of R = 0.998. The electrodes are found to be selective in the presence of other commonly interfering molecules such as ascorbic acid, uric acid, dopamine and acetamidophenol. Thus a nonenzymatic sensor with good selectivity and sensitivity towards glucose in neutral medium has been developed.





