Vural Özdemir Ph.D.
Sanjeeva Srivastava Ph.D.
 Sanjeeva Srivastava, Ph.D.
Sanjeeva Srivastava, Ph.D.
Assistant Professor, Proteomics Lab, IIT-Bombay, India
Identification of Potential Early Diagnostic Biomarkers for Gliomas and Various Infectious Diseases using Proteomic Technologies
The spectacular advancements achieved in the field of proteomics research during the last decade have propelled the growth of proteomics for clinical research. Recently, comprehensive proteomic analyses of different biological samples such as serum or plasma, tissue, CSF, urine, saliva etc. have attracted considerable attention for the identification of protein biomarkers as early detection surrogates for diseases (Ray et al., 2011). Biomarkers are biomolecules that can be used for early disease detection, differentiation between closely related diseases with similar clinical manifestations as well as aid in scrutinizing disease progression. Our research group is performing in-depth analysis of alteration in human proteome in different types of brain tumors and various pathogenic infections to obtain mechanistic insight about the disease pathogenesis and host immune responses, and identification of surrogate protein markers for these fatal human diseases.
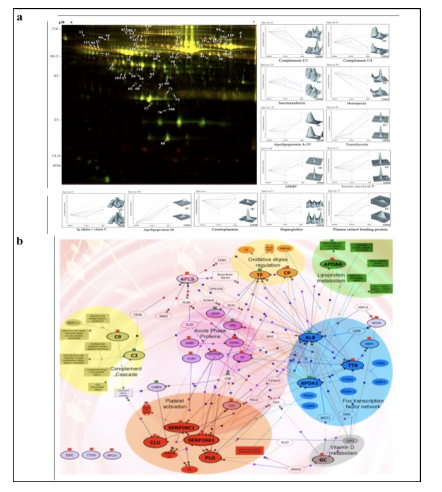
Applying 2D-DIGE in combination with MALDI-TOF/TOF MS we have analyzed the serum and tissue proteome profiles of glioblastoma multiforme; the most common and lethal adult malignant brain tumor (Gollapalli et al., 2012) (Figure 1). Results obtained were validated by employing different immunoassay-based approaches. In serum proteomic analysis we have identified some interesting proteins like haptoglobin, ceruloplasmin, vitamin-D binding protein etc. Moreover, proteomic analysis of different grades (grade-I to IV) of gliomas and normal brain tissue was performed and differential expressions of quite a few proteins such as SIRT2, GFAP, SOD, CDC42 have been identified, which have significant correlation with the tumor growth. While proteomic analysis of cerebrospinal fluid from low grade (grade I & II) vs. high grade (grade III & IV) gliomas revealed modulation of CSF levels of apolipoprotein E, dickkopf related protein 3, vitamin D binding protein and albumin in high grade gliomas. The prospective candidates identified in our studies provide a mechanistic insight of glioma pathogenesis and identification of potential biomarkers. We are also studying the role of JAK/STAT interactome and therapeutic potential of STAT3 inhibitors in gliomas using proteomics approach. Several candidates of the JAK/STAT interactome were identified with altered expression and a significant correlation was observed between STAT3 and PDK1 transcript expression level.
We have also investigated the changes in human serum proteome in different infectious diseases including falciparum and vivax malaria (Ray et al., 2012a; Ray et al., 2012b), dengue (Ray et al., 2012c) and leptospirosis (Srivastava et al., 2012). Although, quite a few serum proteins were found to be commonly altered in different infectious diseases and might be a consequence of inflammation mediated acute phase response signaling, uniquely modulated candidates were identified in each pathogenic infection indicating the some inimitable responses. Further, a panel of identified proteins consists of six candidates; serum amyloid A, hemopexin, apolipoprotein E, haptoglobin, retinol-binding protein and apolipoprotein A-I was used to build statistical sample class prediction models employing PLSDA and other classification methods to predict the clinical phenotypic classes and 91.37% overall prediction accuracy was achieved (Figure 2). ROC curve analysis was carried out to evaluate the individual performance of classifier proteins. The excellent discrimination among the different disease groups on the basis of differentially expressed proteins demonstrates the potential diagnostic implications of this analytical approach.
Keywords: Diagnostic biomarkers, Gliomas, Infectious Diseases, Proteomics, Serum proteome
Acknowledgments: This disease biomarker discovery research was supported by Department of Biotechnology, India grant (No. BT/PR14359/MED/30/916/2010), Board of Research in Nuclear Sciences (BRNS) DAE young scientist award (2009/20/37/4/BRNS) and a startup grant 09IRCC007 from the IIT Bombay. The active support from Advanced Center for Treatment Research and Education in Cancer (ACTREC), Tata Memorial Hospital (TMH), and Seth GS Medical College and KEM Hospital Mumbai, India in clinical sample collection process is gratefully acknowledged.
References :
- Ray S, Reddy PJ, Jain R, Gollapalli K. Moiyadi A, Srivastava S. Proteomic technologies for the identification of disease biomarkers in serum: advances and challenges ahead. Proteomics 11: 2139-61, 2011.
- Gollapalli K, Ray S, Srivastava R, Renu D, Singh P, Dhali S, Dikshit JB, Srikanth R, Moiyadi A, Srivastava S. Investigation of serum proteome alterations in human glioblastoma multiforme. Proteomics 12(14): 2378-90, 2012.
- Ray S, Renu D, Srivastava R, Gollapalli K, Taur S, Jhaveri T, Dhali S, Chennareddy S, Potla A, Dikshit JB, Srikanth R, Gogtay N, Thatte U, Patankar S, Srivastava S. Proteomic investigation of falciparum and vivax malaria for identification of surrogate protein markers. PLoS One 7(8): e41751, 2012a.
- Ray S, Kamath KS, Srivastava R, Raghu D, Gollapalli K, Jain R, Gupta SV, Ray S, Taur S, Dhali S, Gogtay N, Thatte U, Srikanth R, Patankar S, Srivastava S. Serum proteome analysis of vivax malaria: An insight into the disease pathogenesis and host immune response. J Proteomics 75(10): 3063-80, 2012b.
- Srivastava R, Ray S, Vaibhav V, Gollapalli K, Jhaveri T, Taur S, Dhali S, Gogtay N, Thatte U, Srikanth R, Srivastava S. Serum profiling of leptospirosis patients to investigate proteomic alterations. J Proteomics 76: 56-68, 2012.
- Ray S, Srivastava R, Tripathi K, Vaibhav V, Srivastava S. Serum proteome changes in dengue virus-infected patients from a dengue-endemic area of India: towards new molecular targets? OMICS 16(10): 527-36, 2012c.
* Correspondence: Dr. Sanjeeva Srivastava, Department of Biosciences and Bioengineering, IIT Bombay, Mumbai 400 076, India: E-mail: sanjeeva@iitb.ac.in; Phone: +91-22-2576-7779, Fax: +91-22-2572-3480
![Figure 2 (a) Western blot analysis of haptoglobin (HP), serum amyloid A (SAA), and clusterin (CLU) from serum samples of healthy control (HC) [n = 12], falciparum malaria (FM) [n = 12], vivax malaria (VM) [n = 12], Leptospirosis (Lep) [n = 6], dengue fever [DF] [n = 6] and non infectious disease control (NIDC:GBM) [n = 12]. Representative blots of the target proteins are depicted along with their respective relative abundance volumes (volume X 104). All the data are represented as mean ± SE. (b) Discrimination of malaria from dengue, leptospirosis and GBM using PLS-DA analysis. PLS-DA scores Plot for FM (blue spheres, n = 8), VM (green spheres, n = 8), DF (red spheres, n = 6), Lep (grey spheres, n = 6) and GBM (brown spheres, n = 8) samples based on 6 differentially expressed proteins (serum amyloid A, hemopexin, apolipoprotein E, haptoglobin, retinol-binding protein and apolipoprotein A-I) identified using DIGE. The axes of the plot indicate PLSDA latent variables t0-t2.](http://www.amritabioquest.org/conference/2013/wp-content/uploads/sites/2/2015/02/sanjeeva-2.jpg)
 Suryaprakash Sambhara, DVM, Ph.D
Suryaprakash Sambhara, DVM, Ph.D
Chief, Immunology Section, Influenza Division, CDC, Atlanta, USA
Making sense of pathogen sensors of Innate Immunity: Utility of their ligands as antiviral agents and adjuvants for vaccines.
Currently used antiviral agents act by inhibiting viral entry, replication, or release of viral progeny. However, recent emergence of drug-resistant viruses has become a major public health concern as it is limiting our ability to prevent and treat viral diseases. Furthermore, very few antiviral agents with novel modes of action are currently in development. It is well established that the innate immune system is the first line of defense against invading pathogens. The recognition of diverse pathogen-associated molecular patterns (PAMPs) is accomplished by several classes of pattern recognition receptors (PRRs) and the ligand/receptor interactions trigger an effective innate antiviral response. In the past several years, remarkable progress has been made towards understanding both the structural and functional nature of PAMPs and PRRs. As a result of their indispensable role in virus infection, these ligands have become potential pharmacological agents against viral infections. Since their pathways of action are evolutionarily conserved, the likelihood of viruses developing resistance to PRR activation is diminished. I will discuss the recent developments investigating the potential utility of the ligands of innate immune receptors as antiviral agents and molecular adjuvants for vaccines.

John Stanley, Satheesh Babu, Ramacahandran T and Bipin Nair
Pt-Pd decorated TiO2 nanotube array for the non-enzymatic determination of glucose in neutral medium
Rapidly expanding diabetic population and the complications associated with elevated glycemic levels necessitates the need for a highly sensitive, selective and stable blood glucose measurement strategy. The high sensitivity and selectivity of enzymatic sensors together with viable manufacturing technologies such as screen-printing have made a great social and economic impact. However, the intrinsic nature of the enzymes leads to lack of stability and consequently reduces shelf life and imposes the need for stringent storage conditions. As a result much effort has been directed towards the development of ‘enzyme-free’ glucose sensors (Park et al. 2006). In this paper, a non-enzymatic amperometric sensor for selective and sensitive direct electrooxidation of glucose in neutral medium was fabricated based on Platinum-Palladium (Pt–Pd) nanoparticle decorated titanium dioxide (TiO2) nanotube arrays. Highly ordered TiO2 nanotube arrays were obtained using a single step anodization process (Grimes C A and Mor G K 2009) over which Pt–Pd nanoparticles where electrochemically deposited. Scanning Electron Microscopy (SEM) analysis revealed the diameter of the TiO2 nanotubes to be approximately 40 nm. Elemental analysis after electrochemical deposition confirms the presence of Pt–Pd. Electrochemical characterization of the sensor was carried out using cyclic voltammetry technique (−1.0 to +1.0V) in phosphate buffer saline (PBS) pH 7.4. All further glucose oxidation studies were performed in PBS (pH 7.4). The sensor exhibited good linear response towards glucose for a concentration range of 1 μM to 20mM with a linear regression coefficient of R = 0.998. The electrodes are found to be selective in the presence of other commonly interfering molecules such as ascorbic acid, uric acid, dopamine and acetamidophenol. Thus a nonenzymatic sensor with good selectivity and sensitivity towards glucose in neutral medium has been developed.

Manjunath Joshi, Apoorva Lad, Bharat Prasad Alevoor, Aswath Balakrishnan, Lingadakai Ramachandra and Kapaettu Satyamoorthy
Pathological conditions during Type 2 Diabetes (T2D) are associated with elevated risk for common community acquired infections due to poor glycemic control. Multiple studies have indicated specific defects in innate and adaptive immune function in diabetic subjects. Neutrophils play an important role in eliminating pathogens as an active constituent of innate immune system. Apart from canonically known phagocytosis mechanism, neutrophils are endowed with a unique ability to produce extracellular traps (NETs) to kill pathogens by expelling DNA coated with bactericidal proteins and histone. NETosis is stimulated by diverse bacteria and their products, fungi, protozoans, cytokines, phorbol esters and by activated platelets. Considering deregulation of metabolic and immune response pathways during pathological state of diabetes and NETosis as a potential mechanism for killing bacteria, we therefore, investigated whether hyperglycemic conditions modulate formation of neutrophil NETs and attempted to identify underlying immunoregulatory mechanisms. Freshly isolated neutrophils from normal individuals were cultured in absence or presence of high glucose (different concentrations) for 24 hours and activated with either LPS (2 mg/ml) or PMA (20 ng/ml) or IL-6 (20 ng/ml) for 3 hours. NETs were visualized and quantified by addition of DNA binding dye SYTOX green using fluorescence microscope and fluorimetry. NETs were quantified in Normal and diabetic subjects. Serum IL-6 levels were measured using ELISA technique. NETs bound elasatse were quantified in normal and diabetic subjects in presence or absence of DNase. Bacterial killing assays were performed upon infecting E.coli with activated neutrophils from normal and diabetic subjects. Microscopy and fluorimetry analysis suggested dramatic impairment in NETs formation under high glucose conditions. Extracellular DNA lattices formed in hyperglycemic conditions were short lived and unstable leading to rapid disintegration. Subsequent, time course experiments showed that NETs production was delayed in hyperglycemic conditions. To validate our findings more closely to clinical conditions, we investigated the neutrophil activation and NETs formation in diabetic patients. Upon stimulation with LPS for three hours, neutrophils from diabetic subjects responded weakly to LPS and lesser NETs were formed; whereas, neutrophils from normal individuals showed robust release of NETs. In few patients we found short and imperfect NETs in basal conditions suggesting constitutive activation of neutrophils in diabetic subjects. Interestingly, NETs bound elastase activity was reduced in diabetes subjects when compared to non-diabetic individuals, indicating a dysfunction of one of the important protein component of NETs during diabetes. Neutrophils from diabetic subjects released higher levels of IL-6 without any stimulation suggesting an existence of constitutively activated pro-inflammatory state. IL-6 induced NETs formation and was abrogated by high glucose. Weobserved that glycolysis inhibitor 2-DG resensitize the high glucose attenuated LPS and IL-6 induced NETs. a) NETs are influenced by glucose homeostasis, b) IL-6 as potent inducer of energy dependent NETs formation and c) hyperglycemia mimics a state of constitutively active pro-inflammatory condition in neutrophils leading to reduced response to external stimuli making diabetic subjects susceptible for infections.

Aswath Balakrishnan, Kapaettu Satyamoorthy and Manjunath B Joshi
Introduction
Insulin resistance is a hall mark of metabolic disorders such as diabetes. Reduced insulin response in vasculature leads to disruption of IR/Akt/eNOS signaling pathway resulting in vasoconstriction and subsequently to cardiovascular diseases. Recent studies have demonstrated that inflammatory regulator interleukin-6 (IL-6), as one of the potential mediators that can link chronic inflammation with insulin resistance. Accumulating evidences suggest a significant role of epigenetic mechanisms such as DNA methylation in progression of metabolic disorders. Hence the present study aimed to understand the role of epigenetic mechanisms involved during IL-6 induced vascular insulin resistance and its consequences in cardiovascular diseases.
Materials and Methods
Human umbilical vein endothelial cells (HUVEC) and Human dermal microvascular endothelial cells (HDMEC) were used for this study. Endothelial cells were treated in presence or absence of IL-6 (20ng/ml) for 36 hours and followed by insulin (100nM) stimulation for 15 minutes. Using immunoblotting, cell lysates were stained for phosphor- and total Akt levels to measure insulin resistance. To investigate changes in DNA methylation, cells were treated with or without neutrophil conditioned medium (NCM) as a physiological source of inflammation or IL-6 (at various concentrations) for 36 hours. Genomic DNA was processed for HPLC analysis for methyl cytosine content and cell lysates were analyzed for DNMT1 (DNA (cytosine-5)-methyltransferase 1) and DNMT3A (DNA (cytosine-5)-methyltransferase 3A) levels using immunoblotting.
Results
Endothelial cells stimulated with insulin exhibited an increase in phosphorylation of Aktser 473 in serum free conditions but such insulin response was not observed in cells treated with IL-6, suggesting chronic exposure of endothelial cells to IL-6 leads to insulin resistance. HPLC analysis for global DNA methylation resulted in decreased levels of 5-methyl cytosine in cells treated with pro-inflammatory molecules (both by NCM and IL-6) as compared to untreated controls. Subsequently, analysis in cells treated with IL-6 showed a significant decrease in DNMT1 levels but not in DNMT3A. Other pro-inflammatory marker such as TNF-α did not exhibit such changes.
Conclusion
Our study suggests: a) Chronic treatment of endothelial cells with IL-6 results in insulin resistance b) Neutrophil conditioned medium and IL-6 decreases methyl cytosine levels c) DNMT1 but not DNMT3a levels are reduced in cells treated with IL-6.
 Vural Özdemir, MD, Ph.D., DABCP
Vural Özdemir, MD, Ph.D., DABCP
Co-Founder, DELSA Global, Seattle, WA, USA
Crowd-Funded Micro-Grants to Link Biotechnology and “Big Data” R&D to Life Sciences Innovation in India
Vural Özdemir, MD, PhD, DABCP1,2*
- Data-Enabled Life Sciences Alliance International (DELSA Global), Seattle, WA 98101, USA;
- Faculty of Management and Medicine, McGill University, Canada;
ABSTRACT
Aims: This presentation proposes two innovative funding solutions for linking biotechnology and “Big Data” R&D in India with artisan small scale discovery science, and ultimately, with knowledge-based innovation:
- crowd-funded micro-grants, and
- citizen philanthropy
These two concepts are new, and inter-related, and can be game changing to achieve the vision of biotechnology innovation in India, and help bridge local innovation with global science.
Background and Context: Biomedical science in the 21(st) century is embedded in, and draws from, a digital commons and “Big Data” created by high-throughput Omics technologies such as genomics. Classic Edisonian metaphors of science and scientists (i.e., “the lone genius” or other narrow definitions of expertise) are ill equipped to harness the vast promises of the 21(st) century digital commons. Moreover, in medicine and life sciences, experts often under-appreciate the important contributions made by citizen scholars and lead users of innovations to design innovative products and co-create new knowledge. We believe there are a large number of users waiting to be mobilized so as to engage with Big Data as citizen scientists-only if some funding were available. Yet many of these scholars may not meet the meta-criteria used to judge expertise, such as a track record in obtaining large research grants or a traditional academic curriculum vitae. This presentation will describe a novel idea and action framework: micro-grants, each worth $1000, for genomics and Big Data. Though a relatively small amount at first glance, this far exceeds the annual income of the “bottom one billion” – the 1.4 billion people living below the extreme poverty level defined by the World Bank ($1.25/day).
We will present two types of micro-grants. Type 1 micro-grants can be awarded through established funding agencies and philanthropies that create micro-granting programs to fund a broad and highly diverse array of small artisan labs and citizen scholars to connect genomics and Big Data with new models of discovery such as open user innovation. Type 2 micro-grants can be funded by existing or new science observatories and citizen think tanks through crowd-funding mechanisms described herein. Type 2 micro-grants would also facilitate global health diplomacy by co-creating crowd-funded micro-granting programs across nation-states in regions facing political and financial instability, while sharing similar disease burdens, therapeutics, and diagnostic needs. We report the creation of ten Type 2 micro-grants for citizen science and artisan labs to be administered by the nonprofit Data-Enabled Life Sciences Alliance International (DELSA Global, Seattle: http://www.delsaglobal.org). Our hope is that these micro-grants will spur novel forms of disruptive innovation and life sciences translation by artisan scientists and citizen scholars alike.
Address Correspondence to:
Vural Özdemir, MD, PhD, DABCP
Senior Scholar and Associate Professor
Faculty of Management and Medicine, McGill University
1001 Sherbrooke Street West
Montreal, Canada H3A 1G5








